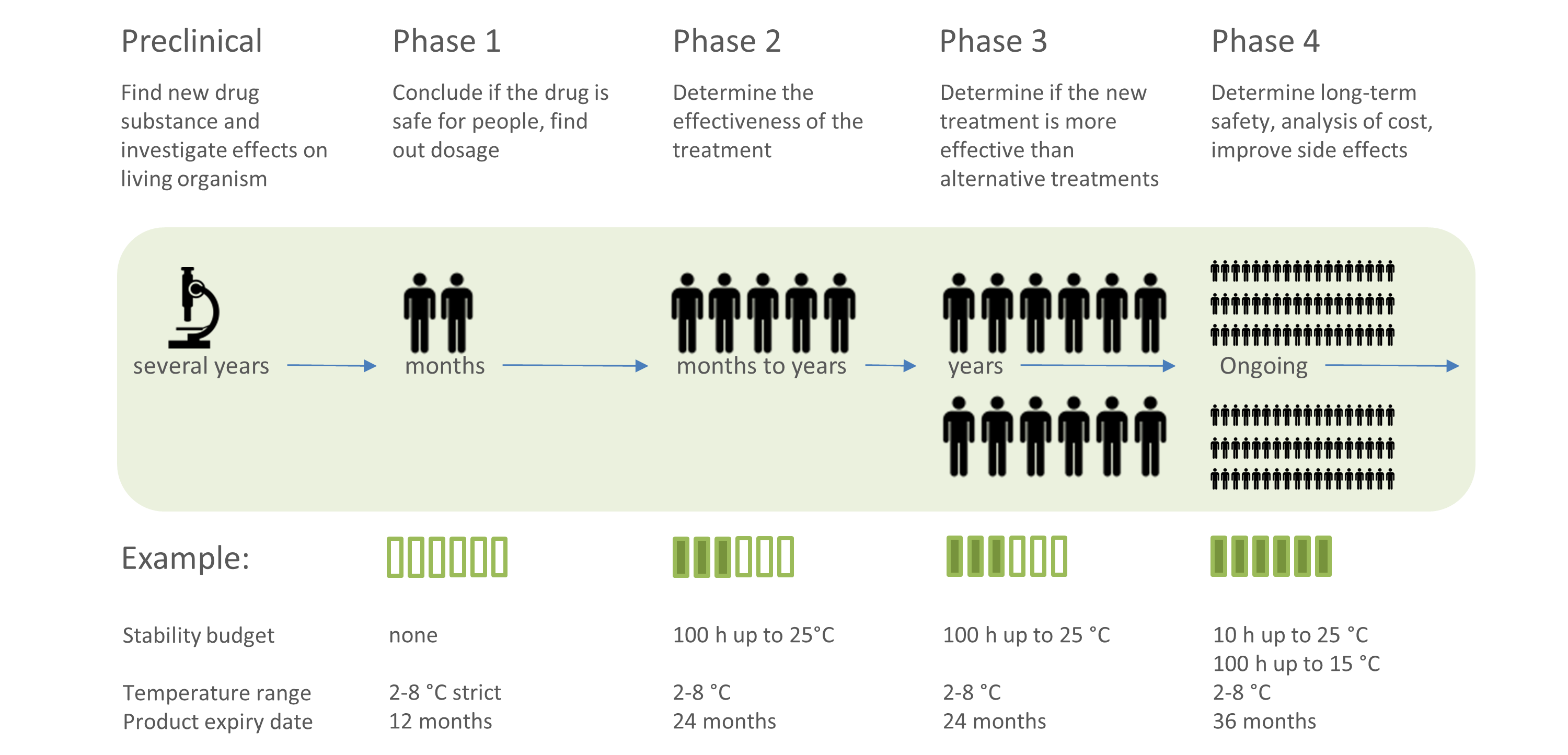
Basics of Cold Chain Monitoring
Find out more on how to manage your temperature-controlled supply chain. Get a general introduction of cold chain monitoring and discover information on topics like stability budget, mean kinetic temperature (MKT), and more!